Ben Feringa’s Nobel Prize-winning molecular motors have received an update, allowing them to both emit light and rotate, Science Advances reports.
The molecular motors from Ben Feringa’s laboratory remain fascinating, yet they also run into a few problems. To make the rotating mechanism work, you often need high-intensity light (usually UV light) which can be harmful to the motor itself or the environment around it. Another point of interest is tracking your motor, as this is not yet possible. Lukas Pfeifer, Nong Hoang and colleagues from the Stratingh Institute for Chemistry and the Zernike Institute for Advanced Materials have tackled these two problems.
The Groningen researchers fabricated three variants of the motor with two light-activated functions in a single molecule. In two of these motors, you can choose which part to activate by different light frequencies. In motor 1, you have two possible wavelengths for rotation: one-photon excitation (1PE) at 445 nm and two-photon excitation (2PE) at near-infrared light of 800 nm. Rotation releases a very short-lived burst of light via photoluminescence. At light of 400 nm you again get 1PE, but instead of rotation you get relatively long-lasting photoluminescence (1.6 ns).
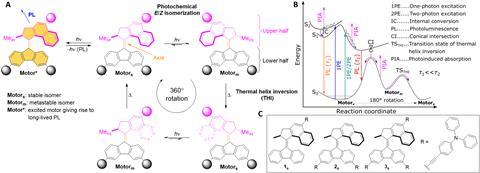
The secret behind this achievement lies in the triphenylamine substituents. The three motors had one, two or three of these substituents, secured with a triple bond. Besides a useful free-electron pair of the nitrogen atom, which you can employ in photochemical reactions, these molecules also increase the cross section (σ2, the measure of the probability of a given interaction between particles).
With this dual action of light emission and rotation, practical biological applications lie in store, the researchers say. Think especially of non-invasive ways to both locate and operate motors in a complex biological system. Or as molecular police cars for a future NanoCar Race (see below).
Pfeifer, L. et al. (2022) Sci. Adv. 8(44), DOI: 10.1126/sciadv.add0410












Nog geen opmerkingen