Egg cells contain a large number of proteins that aid in embryonic development. They store this supply in specific storage complexes, but the exact structure of these complexes remained unclear for a long time. In Nature, researchers from the Hubrecht Institute now present a close-up view of these ‘protein cabinets’.
During the menstrual cycle, an immature egg cell, called an oocyte, matures into an egg cell. In the process, the oocyte grows into one of the largest cells in the female body. To make this possible, the cell accumulates a large number of proteins and other molecules. In the 1960s, scientists discovered that oocytes also contain large fibrous structures, known as cytoplasmic lattices, but their function remained a point of debate. A breakthrough came in 2023, when the team led by cell biologist Melina Schuh at the Max Planck Institute for Multidisciplinary Sciences demonstrated that the grids serve as a storage system for many of the proteins that are crucial for embryonic development.
What the storage complexes consist of and how they are organized remained unclear. Now, the group led by structural biologist Miguel Leung at the Hubrecht Institute has presented their structure in mouse oocytes with unprecedented detail. In doing so, they determined how all the proteins fit together to form the complex. They were not the only ones to do so. Two other publications in Nature and three preprints all demonstrate the same structure, determined in mouse oocytes, eggs and embryos at various stages. According to Leung, it is no coincidence that six research groups converged on this result at the same time: ‘Thanks to advances in structural biology, such as improvements in cryo-EM and image processing, we have now been able to achieve this result.’
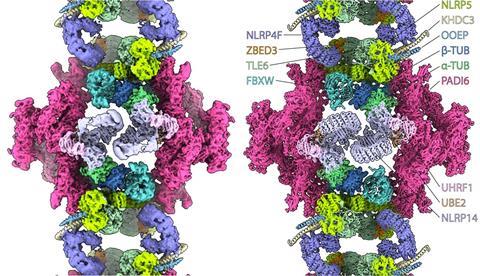
Tubulin storage
To their surprise, the researchers discovered that some proteins in the cytoplasmic grids are ubiquitinating factors—enzymes that regulate protein degradation. It is not yet clear what these proteins are doing there. ‘Perhaps this allows protein levels to increase in the oocyte’, says Leung. ‘Suppose these factors were floating around freely; they could tag proteins that would then be degraded when that is not desirable. By keeping them within the cytoplasmic lattice, this may help the oocyte build up a large number of proteins.’
Another unexpected discovery is that the grids contain tubulin, a protein that helps build the cytoskeleton and transport materials. ‘Tubulin has never before been seen in this form within a protein complex in any cell type. Our hypothesis is that the tubulin can later be released to rapidly polymerize into microtubules for the many cell divisions in the embryo.’
Cell break-in
The researchers used cryo-EM to visually unravel the cytoplasmic grids. For this, they collaborated with cryo-EM expert Willem Noteborn at the Netherlands Centre for Electron Nanoscopy. What proved challenging is that an oocyte (~100 micrometers) is many times larger than the thickness of a cryo-EM sample (~100 nanometers). Leung: ‘The trick was to burst open the oocyte with a detergent, quickly spread the contents over the EM grid, and freeze it immediately.’
Amid the messy cell contents, the researchers then used the microscope to search for the cytoplasmic grids, which were clearly recognizable due to their size. They recorded 40,000 video clips, which allowed them to show that the cytoplasmic grid consists of at least thirteen different proteins, many of which themselves play a crucial role in embryonic development. The protein lattices thus turned out to be composed of the stored proteins themselves.
To confirm these results, the researchers collaborated with the Proteomics team at the Centre for Molecular Medicine at UMC Utrecht. Using mass spectrometry, they demonstrated that the proteins in the cryo-EM reconstruction are also present in large quantities within the oocytes.

Evolution
As a next step, the researchers want to determine how these complex storage structures assemble. Leung is particularly fascinated by how these grids differ across the animal kingdom. ‘All six publications show the structure of the lattices in a mouse. But this differs quite a bit from that of a human. For example, one of the proteins in the lattice of a mouse oocyte isn’t present in ours.’
By determining the implications of these kinds of differences, Leung hopes to gain insight into the various mechanisms underlying the early development of life. ‘Many of the proteins that are important in cytoplasmic lattices in mammals aren’t even found in frogs and fish. How, then, do they develop an embryo? I find this evolutionary aspect very interesting.’
Kılıç, Z.I., van Loenhout, J., Chaillet, M. et al., Cytoplasmic lattices are megadalton storage complexes in mammalian oocytes, Nature (2026), doi:10.1038/s41586-026-10513-8




Nog geen opmerkingen