Sam Stupp’s dancing molecules heal spinal cord injuries
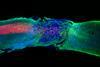
Samuel Stupp designs and develops supramolecular polymers for various applications, ranging from clean energy technology to regenerative medicine. But his polymers are most famous for enabling mice to use their legs again after a spinal cord injury.
From the downtown Chicago office of Samuel Stupp, professor of Materials Science and Engineering, Chemistry, Medicine, and Biomedical Engineering at Northwestern University, you look out onto the Northwestern Memorial Hospital. An inspiring view, if you ask Stupp, who likes to combine scientific fields and cross the boundaries of chemistry to achieve what he set out to do: making a real impact on people’s life.
Want to read more?
Create a free account today!
- Gain access to all our content on chemistry, life sciences and process technology;
- Get our weekly newsletter so you never miss a story.
As a member of the KNCV, KVCV, NBV, or NVBMB you have unlimited access. Log in here.





