Bacteria swim with flagella, and a clever switching system allows them to aim at a target. New research shows in more detail how the flagellar drive changes direction.
Many species of bacteria have one or more flagella. These long whip-like tails that rotate on their own axis allow them to move around, searching for nutrients in the environment or a victim to infect. The flagellum can quickly change its direction of rotation, which determines the bacterium’s course. Exactly how this switch works has been the subject of research for years.
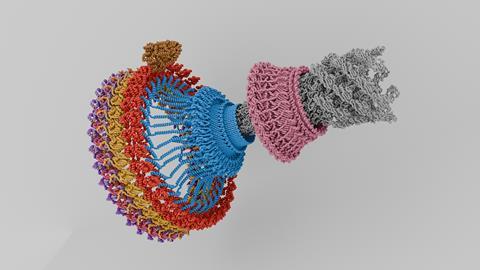
In fact, flagella are a wonderful example of bio-nanotechnology because each flagellum is attached to a rotor: a ring-shaped, rotating protein complex with dozens of subunits. This enables the bacterial cell to convert the flow of protons via motor proteins into counterclockwise or clockwise rotation. The question is how exactly the rotor changes direction.
Two papers recently published in Nature Microbiology provide a clearer picture. A team led by Susan Lea from the UK and Tina Iverson from the US used an electron microscope to zoom in on the switching mechanism. They show how a small change in the shape of a protein determines the direction of rotation.
Gears
‘I think the main conclusion of both papers is very similar’, says Ariane Briegel, professor of electron cryotomography and bacterial chemotaxis at Leiden University. ‘Lea’s paper looks at the whole protein complex, Iverson’s paper analyses a smaller part. But they provide roughly the same model of how the switch might work.’
In simple terms, Lea and Iverson describe a system with two gears. You can think of the ring of the flagel rotor as a large gear driven by a smaller gear: the motor protein (MotA/B). The shape of the large gear determines where the motor protein makes contact. If the motor protein is on the outside, the rotor rotates counterclockwise; if it is on the inside, the rotor rotates clockwise. In an animation on YouTube (see below), Lea’s group shows how this switch works: the motor protein in colour, the flagellar rotor in grey.
Whether this microscopy-based description is 100 percent correct is not entirely certain, says Briegel. ‘But I think the resolution turned out to be very high and I am actually quite convinced that this model is a good approximation of reality. This makes the research exciting and interesting.’
According to Briegel, the research contributes to the understanding of how bacteria move and find their way. Bacteria have sensors in their cell membrane that pick up chemical signals that the bacteria can translate into a direction of rotation of the flagella: counterclockwise, the cell moves steadily forward; clockwise, the cell tumbles and reorients itself. This creates chemotaxis and allows the bacteria to move in a specific direction.
Briegel: ‘Changing the direction of rotation of the flagellum determines their path. Many bacterial species are able to move using this system. It is also an important mechanism because some bacteria use flagella during infection, including salmonella and cholera bacteria.’
Chemosensors
Briegel’s research focuses on the structure and function of chemosensors in the cell membrane of bacteria. According to Briegel, a great deal is already known about how bacteria translate chemosensory information from their environment into the control of their flagella. In particular, the relatively simple model bacterium E. coli has already been mapped in detail.
The challenge is to characterise the chemosensors of other pathogens, such as the spirochetes that cause Lyme disease or syphilis. ‘We are studying a bacterium from this group that causes gingivitis. Spirochetes have a different structure to other bacteria because their cells are very long and thin, so their chemosensors look different to those of E. coli.’
Understanding chemotaxis may provide a clue to infection control. But as far as we know, there are no clinical trials in this area, says Briegel. ‘You often read about it in grant applications. Of course, it would be nice to be able to blind the chemosensory system of a bacterium, for example, with substances that bind to receptors on the cell surface.’
Also have a look at the video from our colleagues at Chemistry World about this research:












Nog geen opmerkingen